Importance of the evaluation of binders in the fight against mycotoxicosis

I. Global prevalence of mycotoxins
Mycotoxins are toxins produced by fungi that, when ingested by animals, cause considerable effects, such as lower production performance and serious economic losses, followed by relevant clinical symptoms in many cases. In addition, they can reach humans either directly (ingestion of contaminated food) or indirectly (through the consumption of animal products).
Their presence is global, and they are found in greater quantities in broken and/or poor-quality grains with high humidity. In addition, some prevalence studies reveal that approximately 50% of contaminated grain contains at least one mycotoxin above legal and/or harmful limits, 45% contains two or more mycotoxins, and the remaining 5% contains 3 or more above these limits. This is important because mycotoxins act synergistically, that is, they enhance the toxicity of other mycotoxins and, additionally, there are hundreds of types, many of which are not considered on a day-to-day basis in sampling.
Therefore, we are facing a major problem for food safety and animal production and efficiency.
II. Addition of mycotoxin binders in the diet
Food safety is now an issue of greater importance worldwide, so there has been a growing interest in controlling feed and food contamination by fungal toxins (mycotoxins).
The use of mycotoxin binders in feed has proven to be an effective method for preventing such negative effects. The objective of these technologies is to bind to mycotoxins and prevent them from being absorbed by the animal and causing pathological effects and productive affectation.
There are several types of mycotoxin binders in the industry. These are usually differentiated or classified by their base molecule (glucomannans -yeasts-, enzymes and silica polymers, among others) and by their effectiveness in capturing mycotoxins.
Silica-based polymers have a high adsorption capacity, but they also have certain drawbacks, such as high dose, lack of homogeneity, and high variability in the spectrum and adsorption capacity of each type of mycotoxin. Silica polymers, or silicate-based products of different types, are effective depending on their crystallographic structure, that is, how they are atomically configured, which will confer one or another binding properties.
In this line, Biovet S.A. has patented the molecule Silicoglycidol, the first mycotoxin binder in 1988. It is a molecule modified from a selected silicate, which, through a complex process, based on heat treatment and ion exchange, acquires an three-dimensional structure that is optimal for binding mycotoxins.
The process, and therefore the structure of the molecule, gives Silicoglycidol, on the one hand, a large adsorption surface area—which means a low dose is required in the diet—and, on the other hand, a greater ability to bind to mycotoxins via hydrogen bonds. Given the nature of this molecule and the chemical groups of mycotoxins, it is capable of binding via hydrogen bonds to all groups of mycotoxins, thereby preventing any type of mycotoxicosis. Furthermore, hydrogen bonds are the strongest bonds in nature, so mycotoxins do not desorb at any pH in the digestive tract and are eliminated through feces without causing harm as they pass through the animal, that is, without causing irritation of the intestinal mucosa (enteritis) or being absorbed (mycotoxicosis).

Molecule Silicoglycidol
III. Evaluation of the effectiveness of a binder
III.I. In vitro evaluation
A suitable method for the evaluation of an in vitro mycotoxin binder should take into account the following factors:
- The conditions of the digestive tract, since physicochemical conditions vary throughout the digestive system and affect the efficiency of the binder.
- Mycotoxins should not be released along the tract: the system should evaluate not only the adsorption, for example, in the stomach, but the final binding percentage, the adsorption efficiency.
- It should allow the evaluation of binding different mycotoxins, since they behave differently.
- It must consider not only the pH values, but also the components and fluids of each digestive compartment, as well as exposure times to them.
Biovet S.A. has patented a method that meets these requirements, which consists of a simulator of the conditions of the different sections of the digestive tract and that allows evaluating the real effectiveness of the binder at the end of the digestive tract (% binding in the stomach – % release in the intestine).
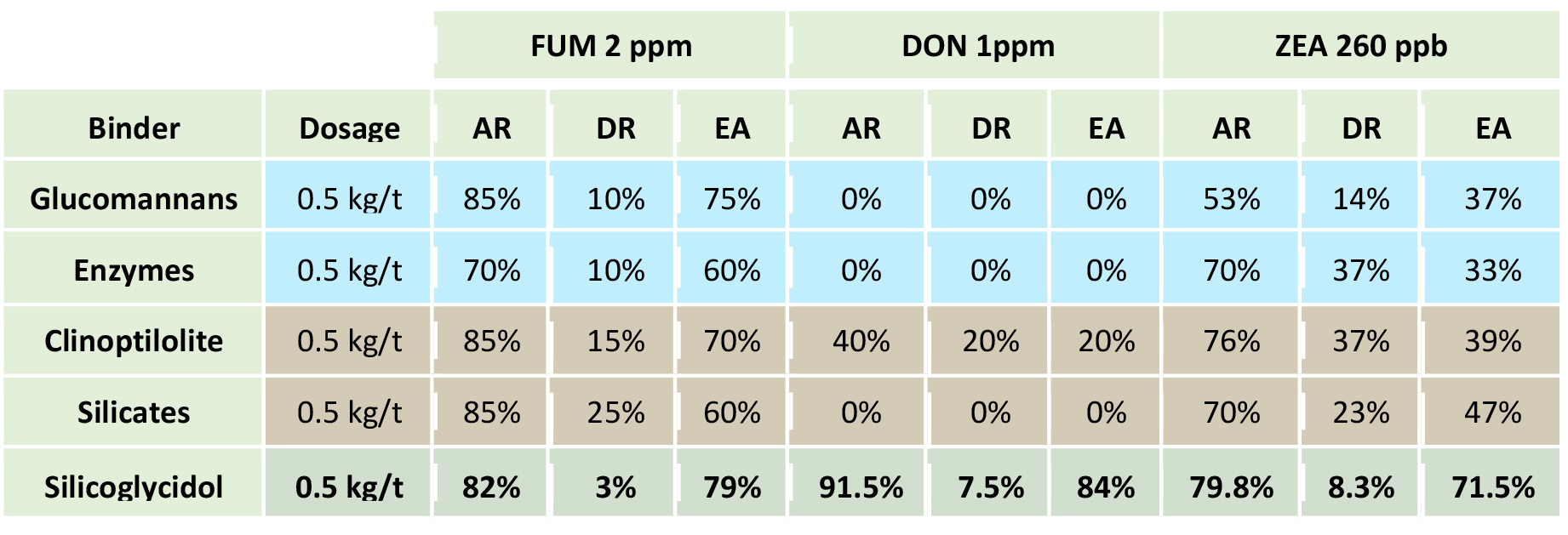
FUM: Fumonisin; DON: deoxinivalenol; ZEA: zearalenone; AR: Adsorption rate at pH 3, DR: desorption rate at pH 9, EA: Adsorption efficiency = AR-DR
In addition, Biovet has recently carried out a trial in collaboration with the University of Helsinki, Finland, also using a simulator of the gastrointestinal tract of birds, based on 3 phases, to which corn contaminated with between 5-10 mg/kg of each mycotoxin is subjected together with the binder:
- Crop phase: considering among other factors: pH 5.5, 40 °C and exposure of 20 minutes.
- Gastric phase: HCl + pepsin, pH 2.5, 40 °C, 90 min, among others.
- Intestinal phase: NaHCO₃ + pancreatin, etc., 40 °C, 3 hours of exposure.

Simulator of the gastrointestinal system developed by the University of Helsinki.
This model replicates the real conditions of the gastrointestinal tract of broilers (crop → stomach → intestine), allowing for the control of pH, temperature and enzymes, among other components, to simulate digestion and interactions between molecules. Additionally, it permits step-by-step observation of toxin binding stability and provides reproducible, ethical, and cost-effective testing before proceeding with in vivo testing. And above all, it ensures that practical results regarding the safety of feed and/or raw materials are accurately reflected.
After each phase, it is centrifuged, filtered (0.22 μm) and then stored (−20 °C), to finally make a reading using LC/Ms-Ms, the gold standard technique, which gives quantitative and qualitative results of capture, as shown in the table below:
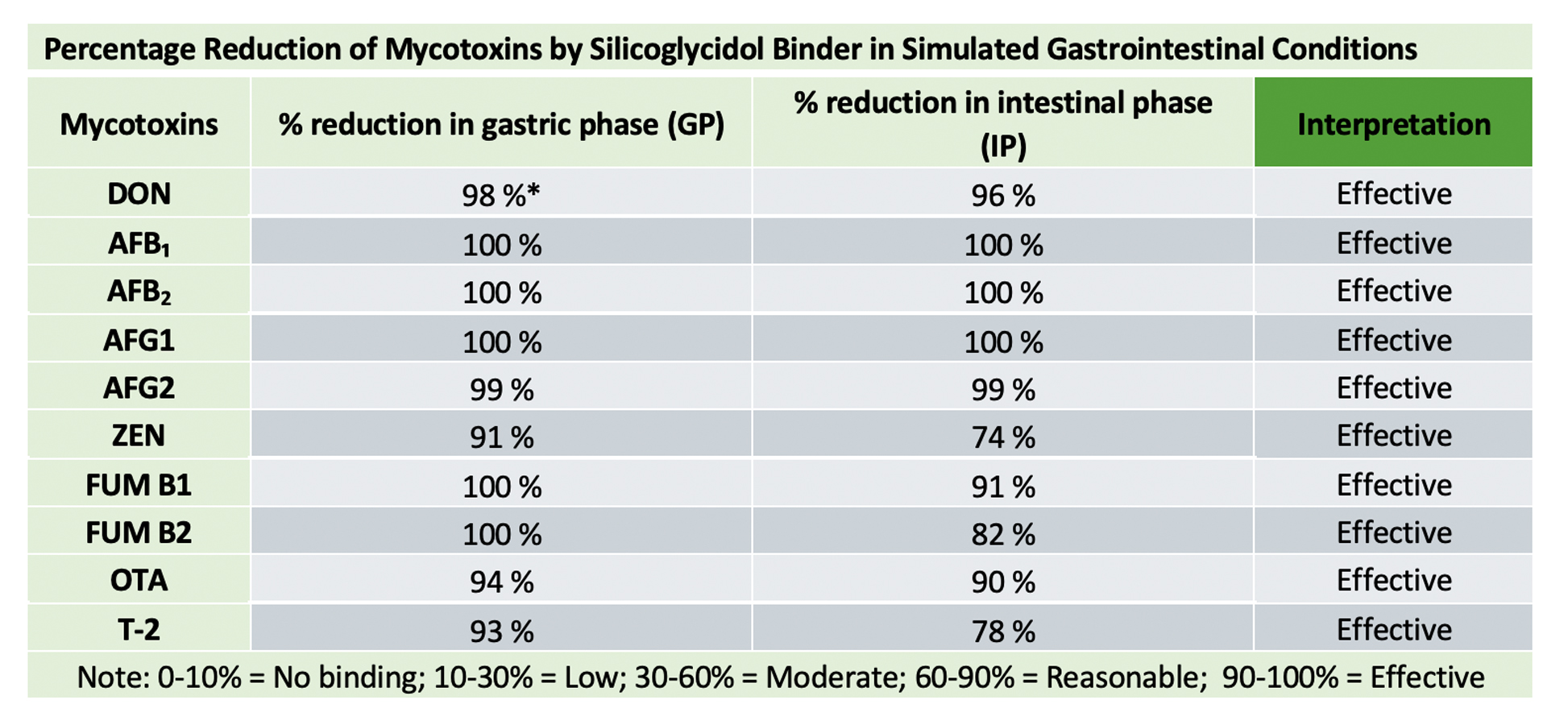
The results showed a high efficacy against mycotoxins of all chemical groups, which confirms a high binding capacity at low doses, and the broad spectrum required to cover any possible type of mycotoxicosis. Furthermore, in parallel trials, it was shown that dietary nutrients, such as vitamins and amino acids, were not adsorbed.
III.II. In vivo evaluation
For the field evaluation of a mycotoxin binder, it is necessary to have at least two batches in the same environmental and handling conditions, and that each of them receives a different binder, that one of the batches is a control, without a binder.
Contaminated feed is administered, and the following are observed:
- Production parameters: they will be improved if the binder is effective.
- Lesions in target organs: necropsies of animals can be performed to study the target organs searching for lesions that coincide with the effects of mycotoxins. On some occasions, the effects are visible without the need to euthanize the animals.
- Relative weight of organs: comparing the weight of an organ with the live weight of the animal allows the efficiency of the organ to be determined (higher relative weight, lower efficiency). Mycotoxins reduce the efficiency of organs (intestine, liver) and thus increase their relative weight.
- More complex parameters, such as the presence of mycotoxin in feces or its metabolites, can also be assessed to calculate how much mycotoxin has been absorbed by the animal.
Field Trial
The objective was to determine the efficiency of Silicoglycidol in the prevention of mycotoxicosis, with a special focus on DON, in broilers, compared to a binder based on a non-optimized silicate.
For the trial, feed contaminated with 275 ppb of DON was administered to the birds and the main production parameters were analyzed
The batches were as follows:
- Control group (Binder): 19773 birds. DON challenge supplemented with a mycotoxin binder at a dose of 3 kg/t, based on a non-optimized silicate.
- Study group (A. Antitox): 50134 birds. DON Challenge supplemented with Alquerfeed Antitox at a dose of 0.5 kg/t
On the results, weight, feed conversion and efficiency, as well as mortality, were analyzed.
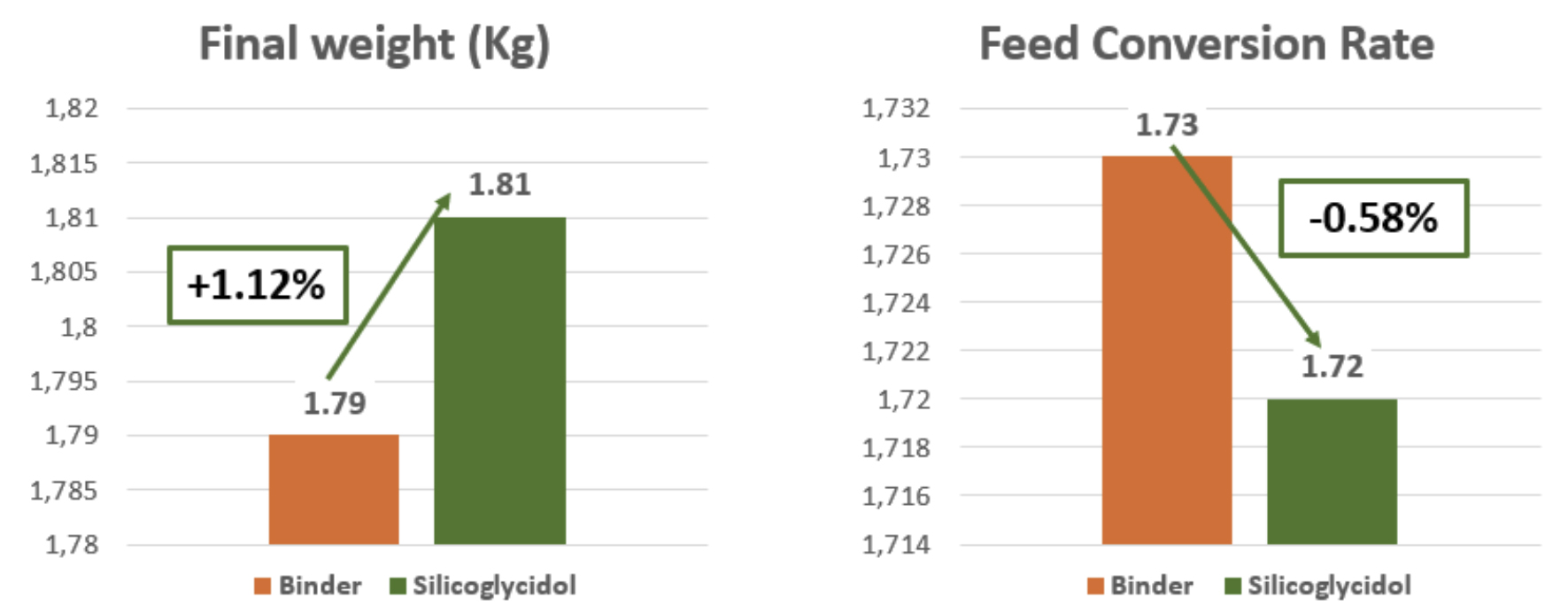
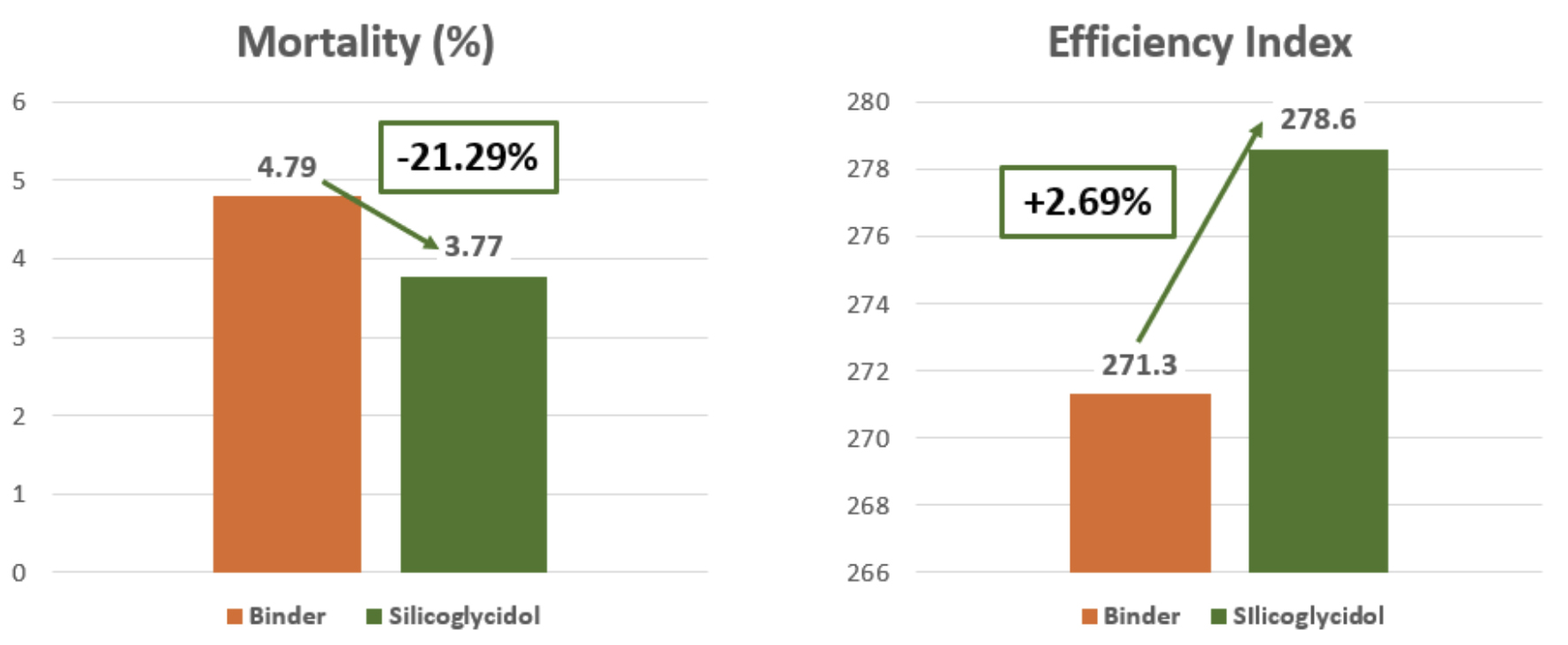
In the batch with Silicoglycidol, an improvement in final weight of 200 grams per bird was observed, which represents a 1.12% increase in weight at the end of the cycle, as well as a reduction of 7 points in the conversion rate. Likewise, the efficiency index improved by 7 points and mortality was reduced by 21% compared to the control batch, below 4.
Therefore, it can be concluded thatSilicoglycidol effectively binds to DON and prevents its absorption and negative effect on broilers, which will have a positive effect on the reproductive parameters of the farm. This is also very significant in economic terms, since it was administered at a dose 6 times lower compared to the other binder.
IV. Conclusions
Mycotoxin binders are an essential tool to avoid the negative effects of these fungal toxins and prevent their entry into products for human consumption.
Evaluation of the binders can be performed in vitro and in vivo. In the first case, models must simulate all conditions of the digestive tract to ensure the efficiency of the binder throughout the tract.
Silicoglycidol, a patented capture molecule, has proven to be effective, both in vitro and on the farm, for the capture of a wide spectrum of mycotoxins, including DON, which causes serious problems in poultry farming and whose overall prevalence is very high.
Silicoglycidol is a technology developed by Biovet, S.A. that is marketed under the trade name Alquerfeed Antitox.